
From Leaf to Lab: How Kratom Is Produced, Processed, and Tested May 2026 By: The Kratom Spot Team Most kratom content covers the basics: leaves get dried, ground into powder, and shipped. What’s rarely documented is the full picture: why harvest timing matters, how drying decisions shape the final product, what fermentation does at a…
 May 20, 2026
May 20, 2026

Kratom Spot Has New Pricing, New Sizes, and a New Way to Save May 2026 By: The Kratom Spot Team At the start of 2026, we made some significant changes to how Kratom Spot is priced and what we offer. If you haven’t heard yet, here’s the full picture — what changed, what’s new, and…
 May 18, 2026
May 18, 2026

The Lab Tested Kratom Buyer’s Checklist (2026 Edition) April 2026 By: Avery Fritz Shopping for kratom in 2026 and comparing products can feel simple, until you look a little closer. Two vendors may sell the same strain name, yet one provides batch records, lab results, and sourcing details you can verify, while the other gives…
 April 28, 2026
April 28, 2026

Traveling With Kratom In 2026: Updated Tips & Considerations April 2026 By: Coco Johnson Planning a trip and wondering about traveling with kratom? In 2026, the answer depends less on “can I pack it?” and more on “where am I going, and what rules apply along the way?” Kratom is not treated the same everywhere,…
 April 21, 2026
April 21, 2026
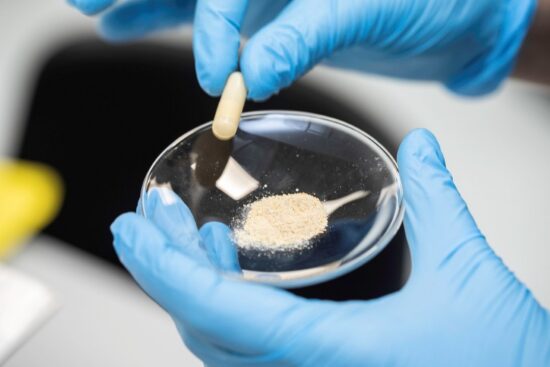
Reading third-party lab tests before your buy kratom. April 2026 By: The Kratom Spot Team If you’ve ever bought coffee from a roaster you trust, you’ve probably seen the little details that build confidence. Information like the origin, roast date, and lot numbers let you verify that the product comes from a trusted, high-quality source….
 April 15, 2026
April 15, 2026

Super Indo vs Ultra Indo vs Indo Standard: Which Fits Your Needs? April 2026 By: Coco Johnson If you’ve ever shopped for Indonesian kratom, you’ve seen the labels multiply fast: “Indo,” “Super Indo,” “Ultra Indo,” sometimes all on the same menu. It can sometimes look like a simple ladder with standard at the bottom, then…
 April 14, 2026
April 14, 2026

How to Use Kratom in Your Daily Routine with Powders, Capsules, & Teas April 2026 By: Avery Fritz When you first hear about Mitragyna speciosa, your first question is often “how to use kratom.” Unfortunately, a lot of how-to guides for beginners jump straight to bold claims or generic one-size-fits-all advice. If you’re looking to…
 April 7, 2026
April 7, 2026

Mitragyna Javanica vs. Kratom: What’s the Difference in 2026? March 2026 By: Coco Johnson If you’ve taken a look at kratom from a variety of angles, you’ve probably seen Mitragyna javanica (or simply “javanica” in product listings or forum threads. Mitragyna javanica is presented as a “kratom cousin,” a “kratom alternative,” or even “kratom-lite.” The…
 March 31, 2026
March 31, 2026

7OH vs Kratom: What is the Difference? March 2026 By: Avery Fritz If you search 7OH vs kratom, you’ll often see people talk about them as if they’re two versions of the same thing, as if 7OH is simply “kratom, but stronger.” However, that statement isn’t accurate. Yet this common confusion between 7OH and kratom…
 March 24, 2026
March 24, 2026

Bentuangie Kratom Guide: Origins, Aroma, and 2026 Buyer Tips March 2026 By: Avery Fritz Some people first notice Bentuangie kratom by the distinctive darker tone of the powder. Others recognize it by aroma alone. Either way, Bentuangie kratom sits in a different lane than many “standard” red, green, or white listings because the name usually…
 March 10, 2026
March 10, 2026

Kratom Allergic Reactions: What You Should Know March 2026 By: Coco Johnson Can you be allergic to kratom? Like any supplement, the answer is yes. People can be allergic to kratom. But there’s a difference between having an allergy and simply not enjoying the effects of a particular kratom strain. A true kratom allergy involves…
 March 3, 2026
March 3, 2026

What does super Indo kratom mean? Learn where this kratom strain comes from, how it’s processed, & choosing the right Super Indo strain for you.
 February 24, 2026
February 24, 2026

How Old Do You Have to be to Buy Kratom? February 2026 By: Coco Johnson Kratom is a popular botanical whose star is on the rise. According to survey data from the National Institute of Health, 5% of the United States population reported using kratom in the past month in 2020. Experts and advocates believe…
 February 17, 2026
February 17, 2026

What Is 7-Hydroxymitragynine? February 2026 By: Avery Fritz 7-hydroxymitragynine is a naturally occurring kratom alkaloid found in Mitragyna speciosa, the tropical tree native to Southeast Asia commonly known as kratom. Among the plant’s more than 40 known alkaloids, 7-hydroxymitragynine—often shortened to 7-OH—is notable for its high potency, even though it occurs in only trace amounts…
 February 14, 2026
February 14, 2026

2026 Kratom Legal Status Update February 2026 By: Coco Johnson If you’ve been hearing snippets of information that leave you asking “is kratom legal,” you’re not alone. In 2026, kratom legality is shifting fast, and the pressure is coming from multiple angles. The community is facing outright state ban bills, agency rulemaking, and proposals that…
 February 12, 2026
February 12, 2026

Inside Kratom Spot: How We Maintain Quality Standards in 2026 February 2026 By: Avery Fritz Kratom (mitragyna speciosa) is soaring in popularity. From powders to capsules to drinks, many people are discovering the powerful wellness benefits of this natural supplement and its alkaloids. However, in the United States, kratom remains unregulated at the federal level….
 February 11, 2026
February 11, 2026

Mitragyna speciosa is a tropical evergreen tree native to Southeast Asia. You may know it more simply as the kratom tree. While kratom has gained attention in the U.S. and Europe over the last decade, the leaves of this tree have been traditionally used in parts of Southeast Asia for generations. What makes Mitragyna speciosa…
 January 23, 2026
January 23, 2026

What Is the Strongest Kratom? A Guide to Quality and Concentration Intro: Reframing “Strength” in Kratom When people search for the “strongest” kratom, they are usually looking for a product that delivers the most impactful experience. However, in the world of high-quality botanicals, “strength” isn’t a one-size-fits-all label. It is a relative term that depends…
 January 10, 2026
January 10, 2026

How to Mix Kratom into Liquids Like a Pro Updated September 2025 By: The Kratom Spot Team SUMMARY: Mixing kratom into liquids is one of the best ways to make it more enjoyable and easier to take. From citrus juices and smoothies to teas, protein shakes, or even chocolate milk, the right liquids and…
 September 30, 2025
September 30, 2025

Traveling with Kratom: What You Need to Know Updated September 2025 By: The Kratom Spot Team SUMMARY: Traveling with kratom in the United States is generally legal, but it’s crucial to understand local laws before hitting the road or boarding a flight. While TSA has no specific ban on kratom, several states and cities do,…
 September 29, 2025
September 29, 2025

Buying Kratom in Bulk: What You Need to Know May 2025 By: The Kratom Spot Team SUMMARY: The blog discusses the benefits of buying kratom in bulk, like cost savings and having a consistent supply, while advising buyers to choose reputable vendors. Discover the importance of product quality, proper storage, and learn about potential legal…
 May 14, 2025
May 14, 2025

Understanding Kratom’s Primary Alkaloid, Mitragynine June 2025 By: The Kratom Spot Team What Is Mitragynine? Mitragynine is the most abundant active compound found in the leaves of Mitragyna speciosa, commonly known as kratom. It belongs to a family of plant-based compounds called alkaloids, which are known for their physiological effects on the human body. In…
 May 6, 2025
May 6, 2025

How Much Kratom Should I Take? A Guide to Kratom Dosing April 2025 By: The Kratom Spot Team SUMMARY: Finding the right kratom dosage is highly individualized and depends on factors like body type, wellness goals, strain, and consumption method. This guide explains dosage ranges, how capsules and powders differ, and provides tips to measure…
 April 30, 2025
April 30, 2025

How to Spot Fake or Low-Quality Kratom Updated September 2025 By: The Kratom Spot Team SUMMARY: Not all kratom on the market is pure or safe, so it’s important to learn how to spot fake or low-quality products. This guide explains the risks of using contaminated kratom, how to identify counterfeit or stale batches, and…
 April 21, 2025
April 21, 2025

What is Trainwreck Kratom? Updated September 2025 By: The Kratom Spot Team SUMMARY: Trainwreck Kratom is a proprietary blend marketed as a “full spectrum” kratom product, but its vague formulation and exaggerated claims make it unreliable and potentially risky. This article explains why Trainwreck may not live up to the hype, highlights safer alternatives, and…
 April 12, 2025
April 12, 2025

























